Sex

48%
48% MALE
52%
52% FEMALE
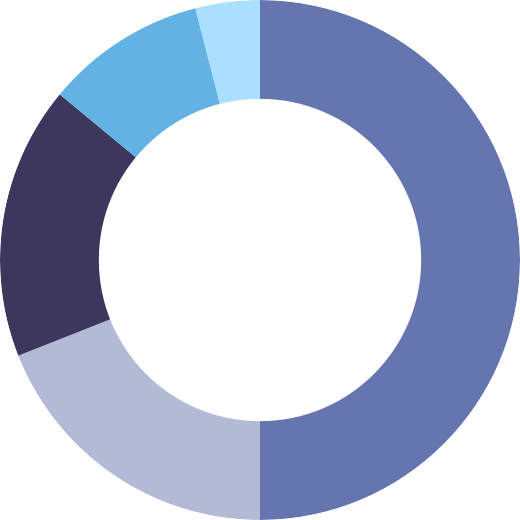
of people in one study were taking at least one mental health medication, including:
ANTIDEPRESSANTS
AT BASELINE
ATYPICAL ANTIPSYCHOTICS
TYPICAL OR COMBINATION ANTIPSYCHOTICS
NO ANTIPSYCHOTICS AT BASELINE

48%
48% MALE
52%
52% FEMALE
10%
10% SCHIZOAFFECTIVE
DISORDER
17%
17% BIPOLAR DISORDER
19%
19% DEPRESSION
4%
4% OTHER
50%
50% SCHIZOPHRENIA
*Once-daily AUSTEDO XR® (deutetrabenazine) extended-release tablets contain the same active ingredient as twice-daily AUSTEDO. Data on this page is based on twice-daily dosing.
Patients can continue on their current psychiatric medication regimen along with the addition of AUSTEDO.
Sign up for TD treatment tips, information about patient
support, and educational resources.
Now Playing:
What to expect with
AUSTEDO® (deutetrabenazine) tablets
Hear professionals discuss their experiences treating Tardive Dyskinesia (TD) with AUSTEDO and what patients can expect when they start treatment.
Video Transcript:
What to Expect with AUSTEDO: Professional Insight
AUSTEDO XR® (deutetrabenazine) extended-release tablets and AUSTEDO® (deutetrabenazine) tablets are prescription medicines that are used to treat adults with movements in the face, tongue, or other body parts that cannot be controlled (tardive dyskinesia).
IMPORTANT SAFETY INFORMATION
AUSTEDO XR and AUSTEDO can cause serious side effects in people with Huntington’s disease, including: depression, suicidal thoughts, or suicidal actions. Do not start taking AUSTEDO XR or AUSTEDO if you are depressed (have untreated depression or depression that is not well controlled by medicine) or have suicidal thoughts. Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts or feelings. This is especially important when AUSTEDO XR or AUSTEDO is started and when the dose is changed. Call your healthcare provider right away if you become depressed, have unusual changes in mood or behavior, or have thoughts of suicide.
Individual results may vary.
Please see the Important Safety Information at the end of this video.
AUSTEDO is a medication that can treat tardive dyskinesia in adults … it’s a tablet that you take twice a day…before we start patients on AUSTEDO, uh, we go over their medication list, so both their prescription drugs, their over-the-counter drugs, any vitamins, supplements they’re on, and any herbal medications they’re taking.
Tardive dyskinesia affects different individuals in different ways. So the overall results may vary greatly from one person to another. As with any treatment, with the use of AUSTEDO, some people may experience side effects, and some people may not necessarily reap benefits associated with the use of the medicine…But when you take a look at the results of the clinical trials, they were actually quite nice in really seeing significant differences for individuals in reducing that movement burden.
Educating patients about what to expect with AUSTEDO, as it correlates with whatever condition it is that we’re treating for them. It’s really an important topic. When they know that they don’t have to compromise with maybe getting worse with whatever it is that we’ve been treating, but, in fact, now also have a remedy for their movement problem, people oftentimes, feel a sense of relief. They oftentimes speak of, again, a sense of feeling like a weight’s been lifted off of them that they didn’t think would be lifted from them previously. So it’s really gratifying being able to let people know of that we have treatments that can help.
Encouraging patients to continue on with their treatment with AUSTEDO® to address their tardive dyskinesia is a very important point. We always wanna reinforce in our patients to comply or to take their medicines as they’re suggested to be taken. That doesn’t always happen. So it’s really important to reinforce that if someone stops taking a medicine like AUSTEDO®, we may well lose ground in that those movements may actually come back.
The way that I encourage patients to stay on the medication is, um, you know, I make it meaningful for them. I find what symptom it is, or what is been improved for them, and ask them, to continue that stability, that they need to continue on, on this medication.
Teva as a company offers resources to help patients start and continue their medication.
APPROVED USE
AUSTEDO® XR (deutetrabenazine) extended-release tablets and AUSTEDO® (deutetrabenazine) tablets are prescription medicines that are used to treat:
the involuntary movements (chorea) of Huntington’s disease. AUSTEDO XR and AUSTEDO do not cure the cause of the involuntary movements, and it does not treat other symptoms of Huntington’s disease, such as problems with thinking or emotions.
movements in the face, tongue, or other body parts that cannot be controlled (tardive dyskinesia).
It is not known if AUSTEDO XR and AUSTEDO are safe and effective in children.
IMPORTANT SAFETY INFORMATION
AUSTEDO XR and AUSTEDO can cause serious side effects in people with Huntington’s disease, including: depression, suicidal thoughts, or suicidal actions. Do not start taking AUSTEDO XR or AUSTEDO if you are depressed (have untreated depression or depression that is not well controlled by medicine) or have suicidal thoughts. Pay close attention to any changes, especially sudden changes, in mood, behaviors, thoughts or feelings. This is especially important when AUSTEDO XR or AUSTEDO is started and when the dose is changed. Call your healthcare provider right away if you become depressed, have unusual changes in mood or behavior, or have thoughts of suicide.
Do not take AUSTEDO XR or AUSTEDO if you:
have Huntington’s disease and are depressed or have thoughts of suicide.
have liver problems.
are taking reserpine. Do not take medicines that contain reserpine with AUSTEDO XR or AUSTEDO. If your healthcare provider plans to switch you from taking reserpine to AUSTEDO XR or AUSTEDO, you must wait at least 20 days after your last dose of reserpine before you start taking AUSTEDO XR or AUSTEDO.
are taking a monoamine oxidase inhibitor (MAOI) medicine. Do not take an MAOI within 14 days after you stop taking AUSTEDO XR or AUSTEDO. Do not start AUSTEDO XR or AUSTEDO if you stopped taking an MAOI in the last 14 days. Ask your healthcare provider or pharmacist if you are not sure.
are taking tetrabenazine. If your healthcare provider plans to switch you from tetrabenazine to AUSTEDO XR or AUSTEDO, take your first dose of AUSTEDO XR or AUSTEDO on the day after your last dose of tetrabenazine.
are taking valbenazine.
Other possible serious side effects include:
Irregular heartbeat (QT prolongation). AUSTEDO XR and AUSTEDO increases your chance of having certain changes in the electrical activity in your heart. These changes can lead to a dangerous abnormal heartbeat. Taking AUSTEDO XR or AUSTEDO with certain medicines may increase this chance.
Neuroleptic Malignant Syndrome. Call your healthcare provider right away and go to the nearest emergency room if you develop these signs and symptoms that do not have another obvious cause: high fever, stiff muscles, problems thinking, very fast or uneven heartbeat, or increased sweating.
Restlessness. You may get a condition where you feel a strong urge to move. This is called akathisia.
Parkinsonism. Symptoms include: slight shaking, body stiffness, trouble moving, trouble keeping your balance, or falls.
Sleepiness (sedation) is a common side effect of AUSTEDO XR and AUSTEDO. While taking AUSTEDO XR or AUSTEDO, do not drive a car or operate dangerous machinery until you know how AUSTEDO XR or AUSTEDO affects you. Drinking alcohol and taking other drugs that may also cause sleepiness while you are taking AUSTEDO XR or AUSTEDO may increase any sleepiness caused by AUSTEDO XR and AUSTEDO.
The most common side effects of AUSTEDO in people with Huntington’s disease include sleepiness (sedation), diarrhea, tiredness, and dry mouth.
The most common side effects of AUSTEDO in people with tardive dyskinesia include inflammation of the nose and throat (nasopharyngitis) and problems sleeping (insomnia).
The most common side effects of AUSTEDO XR are expected to be similar to AUSTEDO in people with Huntington’s disease or tardive dyskinesia.
These are not all the possible side effects of AUSTEDO XR or AUSTEDO. Call your doctor for medical advice about side effects. You are encouraged to report side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
Please read the Medication Guide available at AUSTEDO.com, or by calling 1-800-887-8100.
The information on this site is intended for healthcare professionals in the United States. Are you a healthcare professional in the United States?

